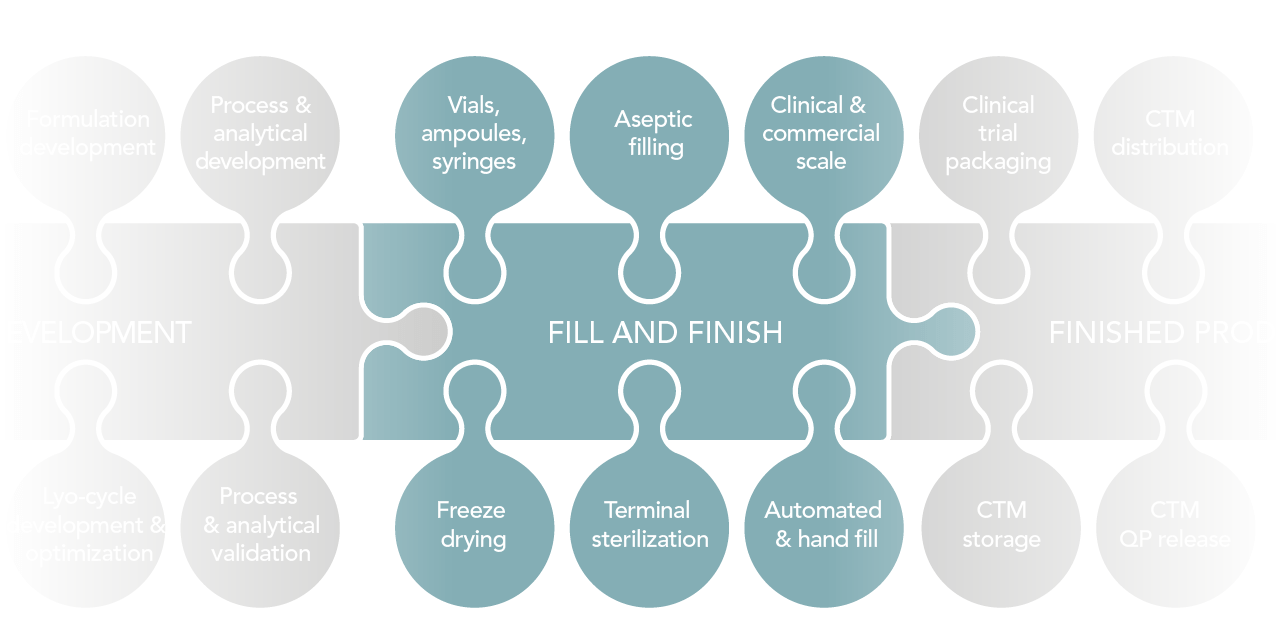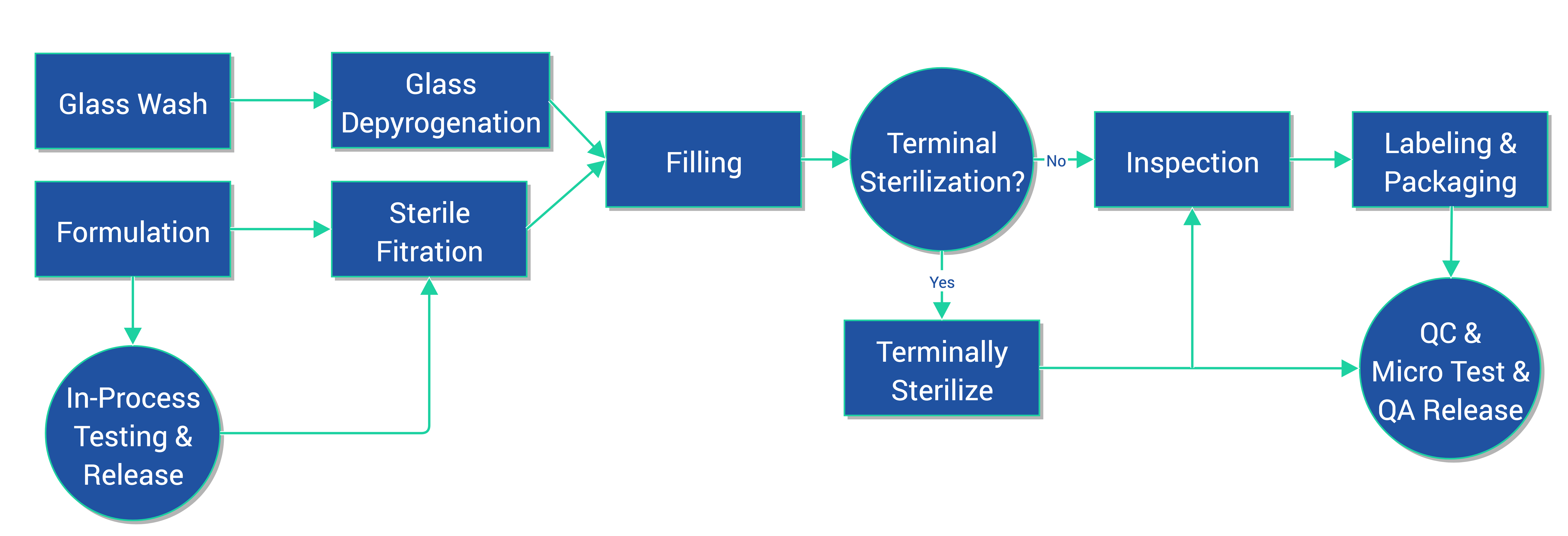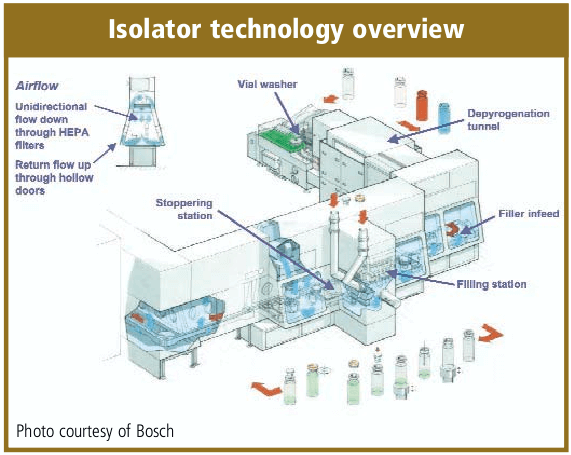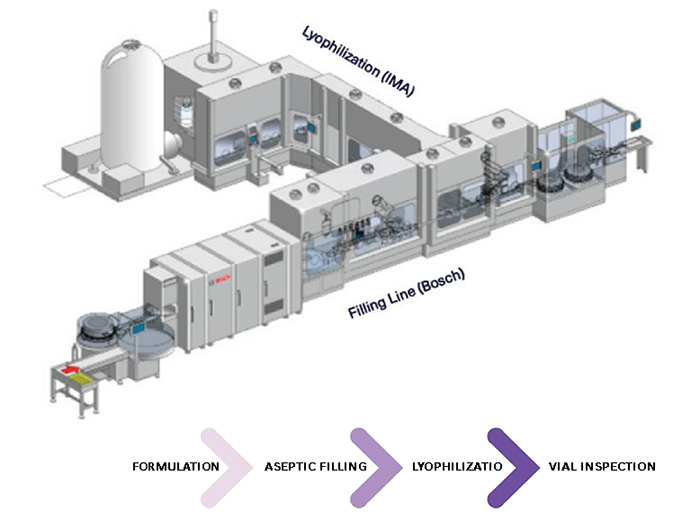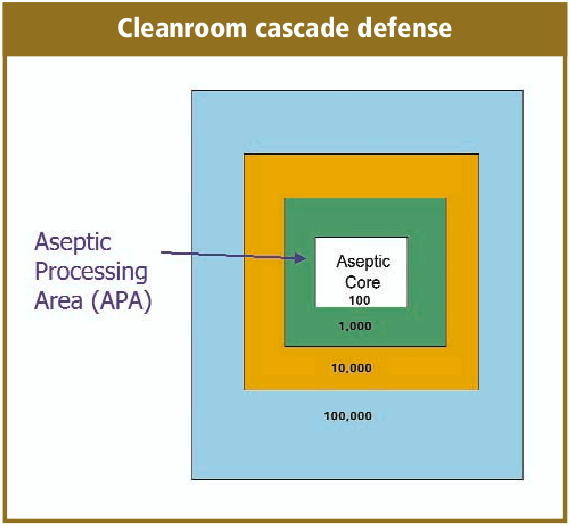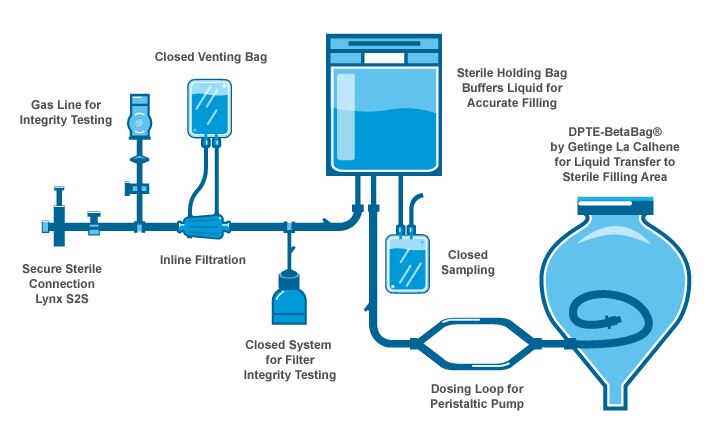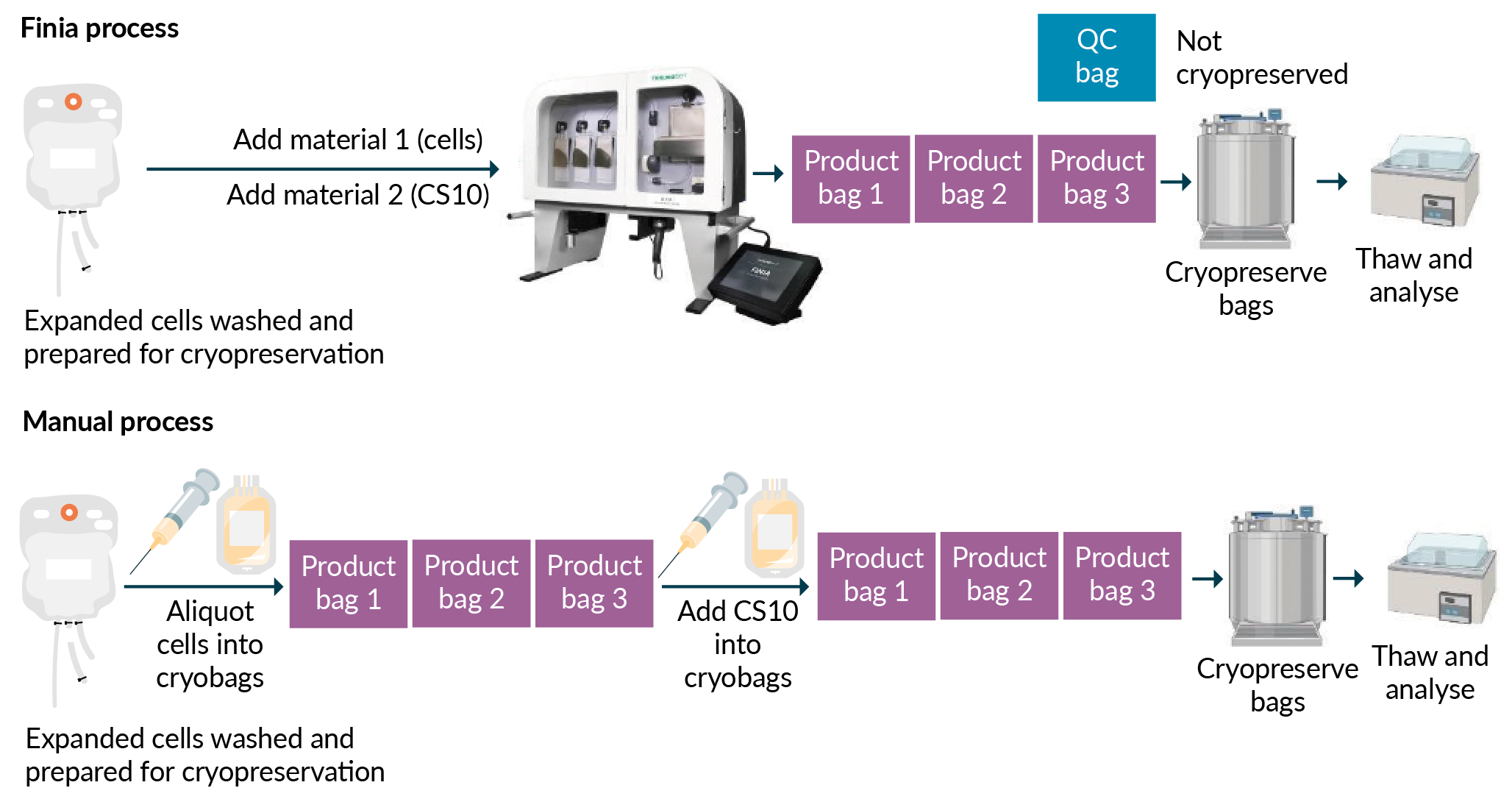
De-risking the final formulation, fill and finish step in cell therapy manufacturing: considerations for an automated solution

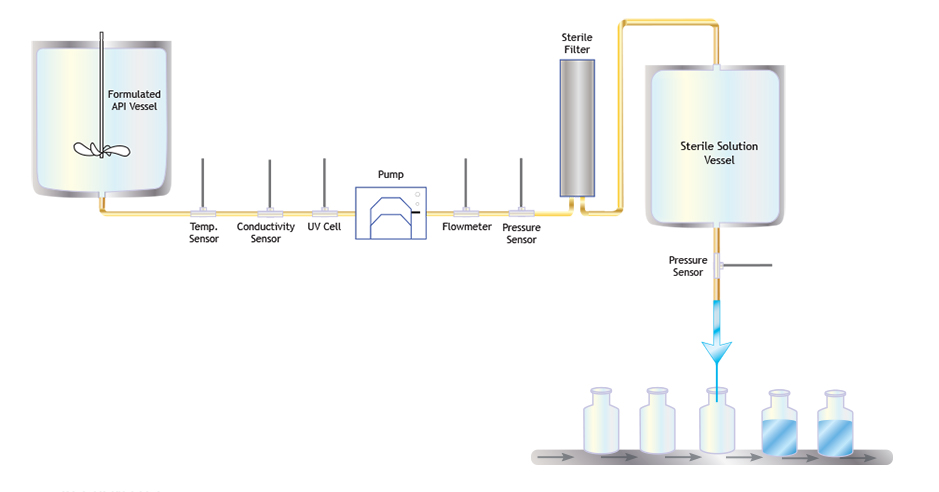
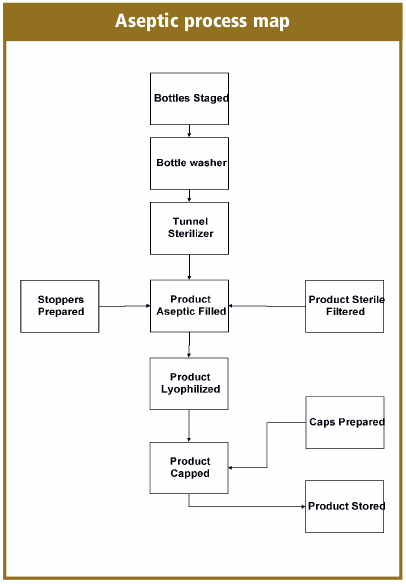
Figure 1 from Optimizing the Design and Operation of Fill-Finish Facilities using Process Simulation and Scheduling Tools by | Semantic Scholar
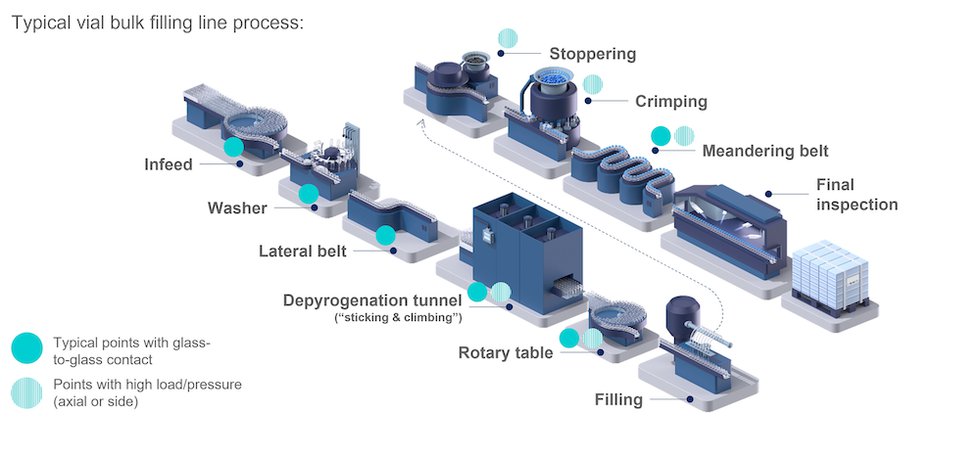
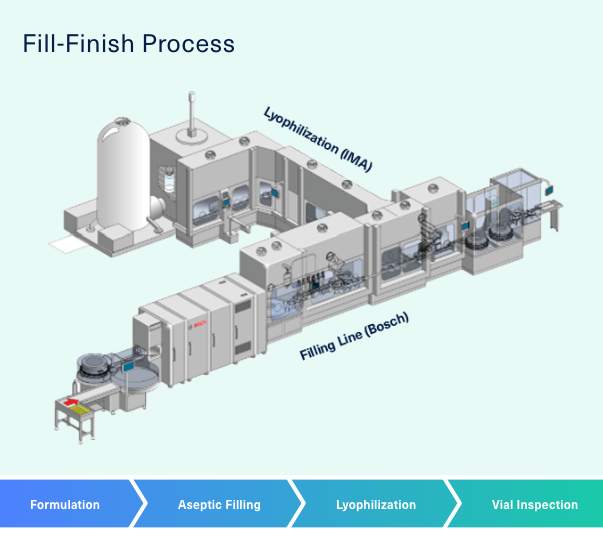
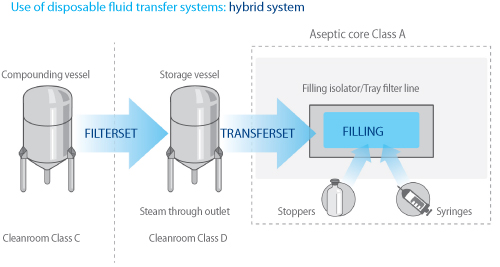
Improved fill-and-finish processes reduce the Total Cost of Ownership - European Pharmaceutical Manufacturer